FUJIFILM Announces Start in USA of Clinical Phase II Trial of Antiviral Drug AVIGAN for COVID-19 Patients
Fujifilm has just issued a press release, where they officially announce that their anti-influenza virus drugs “Avigan ® Tablets” has started a clinical phase II study in patients new coronavirus infection in the United States.
The purpose of this study is to confirm the therapeutic efficacy and safety of Avigan.
The study is scheduled to be conducted at three facilities: Brigham and Women’s Hospital, Massachusetts General Hospital, and Massachusetts State University Medical School.
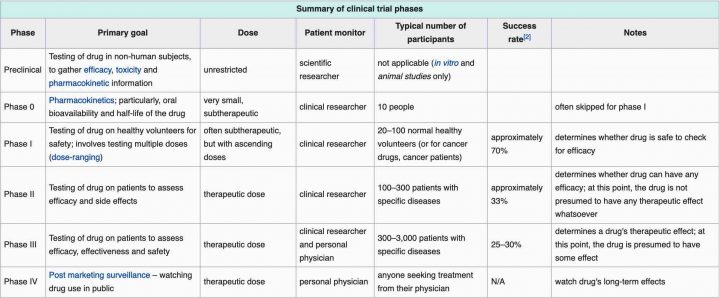
In case you are unaware of the different phases, down below is a table taken from wikipedia.
In Japan, they are at Phase III of the tests already, as we reported here.