FUJIFILM Announces Start in USA of Clinical Phase II Trial of Antiviral Drug AVIGAN for COVID-19 Patients
Fujifilm has just issued a press release, where they officially announce that their anti-influenza virus drugs “Avigan ® Tablets” has started a clinical phase II study in patients new coronavirus infection in the United States.
The purpose of this study is to confirm the therapeutic efficacy and safety of Avigan.
The study is scheduled to be conducted at three facilities: Brigham and Women’s Hospital, Massachusetts General Hospital, and Massachusetts State University Medical School.
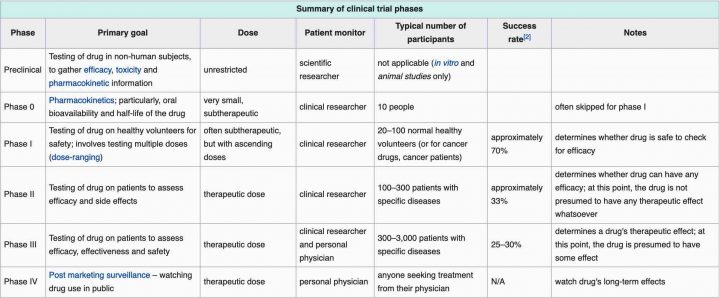
In case you are unaware of the different phases, down below is a table taken from wikipedia.
In Japan, they are at Phase III of the tests already, as we reported here.
Press Release
Fujifilm announces the start of a phase II clinical trial of its influenza antiviral drug “Avigan® Tablet” for COVID-19 patients in the U.S.
TOKYO, April 9, 2020 — FUJIFILM Corporation (President: Kenji Sukeno) announces the initiation of a U.S. phase II clinical trial to evaluate the safety and efficacy of its influenza antiviral drug “Avigan® Tablet” (generic name: favipiravir) for patients with COVID-19, a respiratory infection caused by the novel SARS-CoV-2 coronavirus.
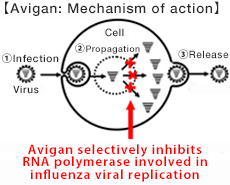
Avigan, approved in Japan for manufacture and sale as an influenza antiviral drug, selectively inhibits RNA polymerase necessary for influenza virus replication. Due to this mechanism of action, it is expected that Avigan may potentially have an antiviral effect on the new coronavirus, because like influenza viruses, coronaviruses are single-stranded RNA viruses that also depend on viral RNA polymerase. On March 31, Fujifilm announced the start of a phase III clinical trial of Avigan for COVID-19 patients in Japan.
The clinical trial in the U.S. will enroll approximately 50 patients with COVID-19, in collaboration with Brigham and Women’s Hospital, Massachusetts General Hospital, and the University of Massachusetts Medical School.
Fujifilm will continue to work to establish a treatment method for COVID-19 patients through conducting clinical trials, and to contribute to ending the spread of this global pandemic as soon as possible by increasing the production of Avigan in collaboration with strategic partners.
[About “Avigan®Tablet”] Avigan Tablet was approved for manufacture and sale in Japan in 2014 as an influenza antiviral drug. The drug is to be considered for use only when there is an outbreak of novel or re-emerging influenza virus infections in which other influenza antiviral drugs are either not effective or insufficiently effective, and the Japanese government decides to use the drug as a countermeasure against such influenza viruses. The drug is to be supplied only at the discretion of Japan’s Health, Labor and Welfare Ministry. It is therefore only manufactured and distributed upon request by the Japanese Government. As such Avigan has never been generally distributed in the market and is not available at hospitals and pharmacies in Japan. Avigan is not approved for distribution in the U.S. or any overseas countries.